Automated Synthesis of Peptoids and Peptoid-Peptide Hybrids
Introduction
Peptoids are polymers of various N-substituted glycines. Though similar in structure to peptides (Figure 1), peptoids are resistant to proteolytic degradation, attributed to the complete substitution of their amide bonds. The increased stability of peptoids in vivo makes them an attractive peptidomimetic target for drug discovery and development.1,2
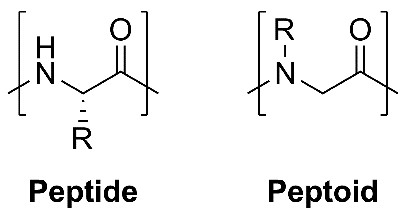
Figure 1. Comparison of peptide and peptoid structure
Peptoids and peptoid-peptide hybrids are typically synthesized through a "sub-monomer" process, which consists of two steps: (1) acylation with bromoacetic acid and N,N’diisopropylcarbodiimide (DIC) and (2) nucleophilic displacement with a monosubstituted amine (Figure 2).1,3
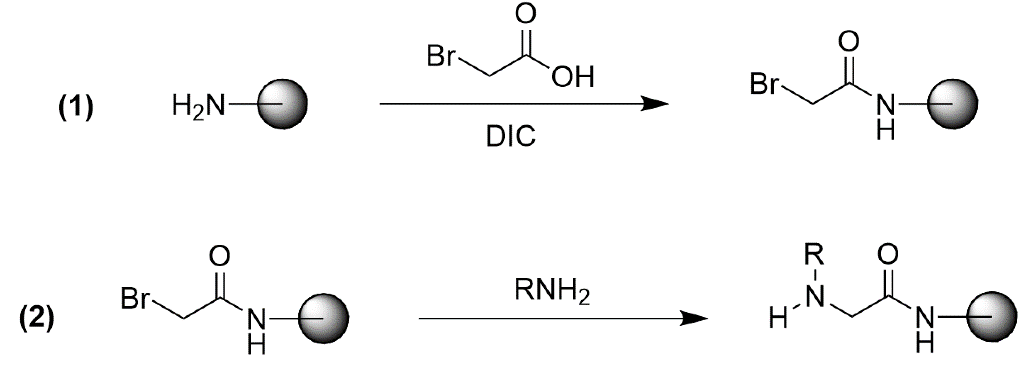
Figure 2. Typical synthesis of peptoids
Because many structurally diverse monosubstituted amines are commercially available, peptoids with a wide variety of side chains can be readily synthesized.2,3 However, conventional synthesis of peptoids can take up to three hours per residue.1 Microwave irradiation has been shown to significantly reduce this time, making production of peptoid libraries and peptoid-peptide hybrids much more viable.1–3
