We use cookies to make your experience better. To comply with the new e-Privacy directive, we need to ask for your consent to set the cookies. Learn more.
Performing Inert Chemistry
Introduction
Microwave heating is employed in numerous synthetic transformations, including nanomaterial assembly, polymerization reactions, and small molecule synthesis.1–3 Nearly any conventionally heated transformation can be adapted for microwave irradiation, including those employing sensitive synthons and transition-metal catalysts.4 Benefits of microwave heating include decreased waste generation, increased product purity, and shortened reaction times.
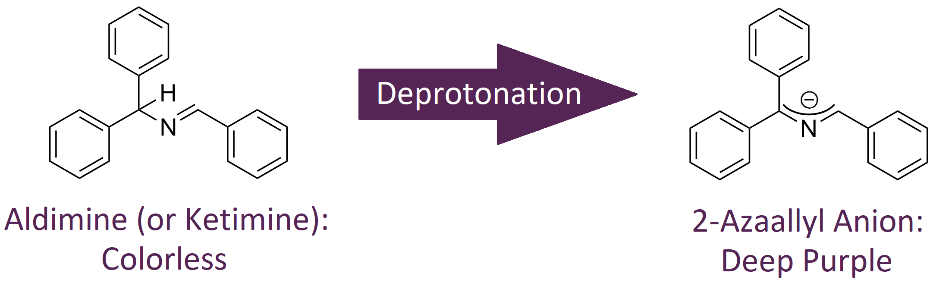
Figure 1: Generation of 2-azaallyl anions from benzhydryl-substituted aldimines (or benzophenone-substituted ketimines)
The elevated reaction rates observed with microwave irradiation enables quick reaction optimization and rapid compound library screening. Productivity is further improved when partnered with autosampler accessories, such as CEM’s Discover® 2.0 with Autosampler 12 or 48, which allows multiple experiments to be prepared simultaneously and then queued to run sequentially. For experiments utilizing sensitive reagents, however, successful autosampler implementation relies on the reaction vessel’s ability to maintain an inert atmosphere while in queue and post-reaction.
To demonstrate the Discover 2.0 10 and 35 mL vessel’s ability to maintain an inert atmosphere, a study employing 2-azaallyl anions was performed. 2-Azaallyl anions, which are generated through deprotonation of benzhydryl-substituted aldimines (and benzophenone-substituted ketimines) (Figure 1), are airsensitive synthons receiving much attention for their utility in amine assembly.5–8 Upon formation, 2-azaallyl anions are vibrantly colored (often purple) and when quenched, turn colorless and transparent (Figure 2). This dramatic color change allows for ready observation of vessel atmospheric conditions.
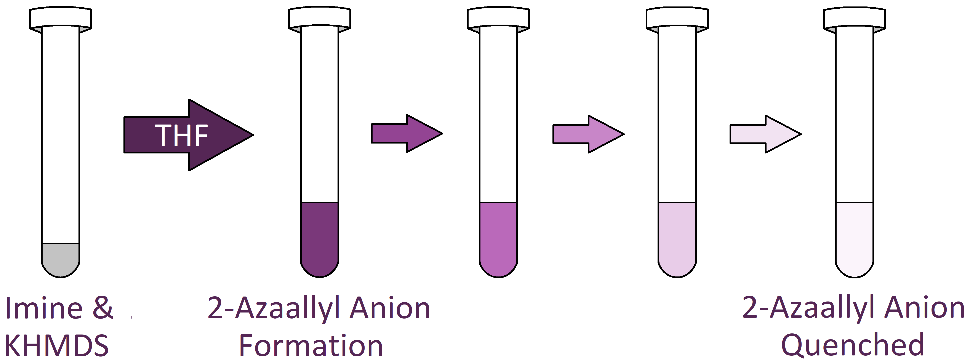
Figure 2: 2-Azaallyl anion solutions are vibrantly colored (often purple) upon formation, turning colorless and transparent when quenched.