Microwave Chemistry with Non-polar Reaction Solutions
Introduction
Microwave heating has been a versatile and powerful tool for synthetic chemists since the 1980s, advancing nanomaterial assembly, drug discovery, peptide synthesis, and more. The two mechanisms by which microwaves generate heat, ionic conduction and dipolar rotation, rely on an ion or molecule’s ability to align itself with the ever-oscillating electric field of a microwave. In general, highly polarized species undergo ionic conduction and dipolar rotation most readily, while low-polarity species are slowest to heat.
A common measure of polarity is the dielectric constant (ε’), which measures a compound’s ability to store electrical charges. Though useful, this value can sometimes contradict another important dielectric parameter, the dielectric loss constant (ε’’), which measures a solvent’s ability to dissipate absorbed microwave energy to its surroundings. In microwave heating, the dielectric loss constant (ε’) provides the best gauge of a material’s ability to efficiently absorb microwaves and undergo heating. Typically, high-absorbing materials have an ε’ greater than 14, while low-absorbing solvents have an ε’ less than 1 (Table 1).
Table 1: The dielectric constants and dielectric loss constants of six common solvents
| Solvent | Dielectric Constant (ε') | Dielectric Loss Constant (ε’’) |
| DMSO | 45.0 | 37.125 |
| Water | 80.4 | 9.889 |
| Acetonitrile | 37.5 | 2.325 |
| DCM | 9.1 | 0.382 |
| Toluene | 2.4 | 0.096 |
| Hexanes | 1.9 | 0.038 |
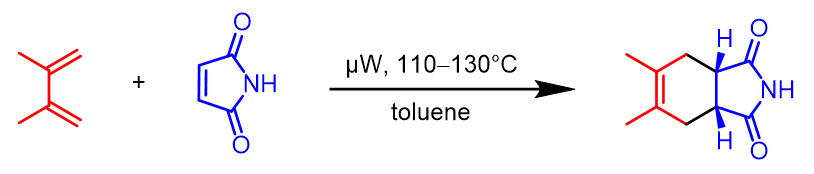
Scheme 1:Microwave-heated Diels-Alder reaction in toluene